Oncology Pipeline
We are a clinical stage biopharmaceutical company focused on the discovery and development of covalent small molecules to treat patients with genetically defined cancers and metabolic diseases. A covalent small molecule is a synthetic compound that forms a permanent bond to its target protein and offers a number of potential advantages over conventional non-covalent drugs, including greater target selectivity, lower drug exposure, and the ability to drive a deeper, more durable response.
We are utilizing our proprietary FUSION™ System to discover, design, and develop a pipeline of next-generation covalent-binding small molecule medicines designed to maximize clinical benefit for patients with various cancers and metabolic diseases, including diabetes.
We aim to cure.
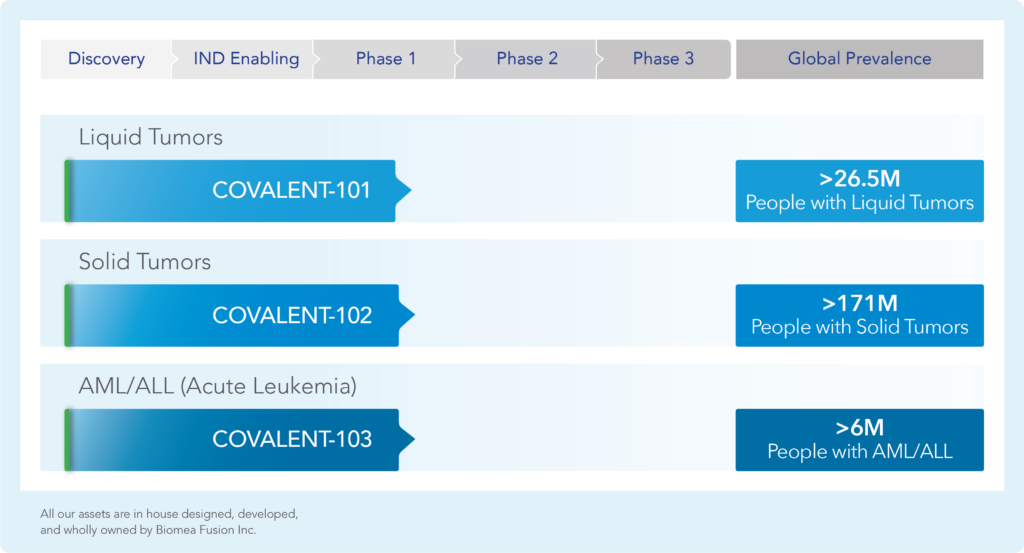
The following table summarizes our product candidate pipeline. We own full worldwide development and commercialization rights to
all of our programs.
BMF-219 – Menin Inhibitor in Oncology
BMF-219 was discovered and developed in-house at Biomea using the company’s proprietary FUSION™ System. BMF-219 is a covalently binding inhibitor of menin, a protein known to play an essential role in oncogenic signaling in genetically defined leukemias as well as in diabetes. Preclinically, BMF-219 has demonstrated in well-established acute leukemia cell lines robust downregulation of key leukemogenic genes in addition to menin itself. Additionally, BMF-219 has shown anticancer activity in multiple in vitro, in vivo, and ex vivo models of acute leukemia, multiple myeloma, diffuse large B-cell lymphoma, and chronic lymphocytic leukemia. BMF-219 is currently being evaluated in clinical trials enrolling patients with specific menin-dependent mutations in liquid and solid tumors.
Clinical Development
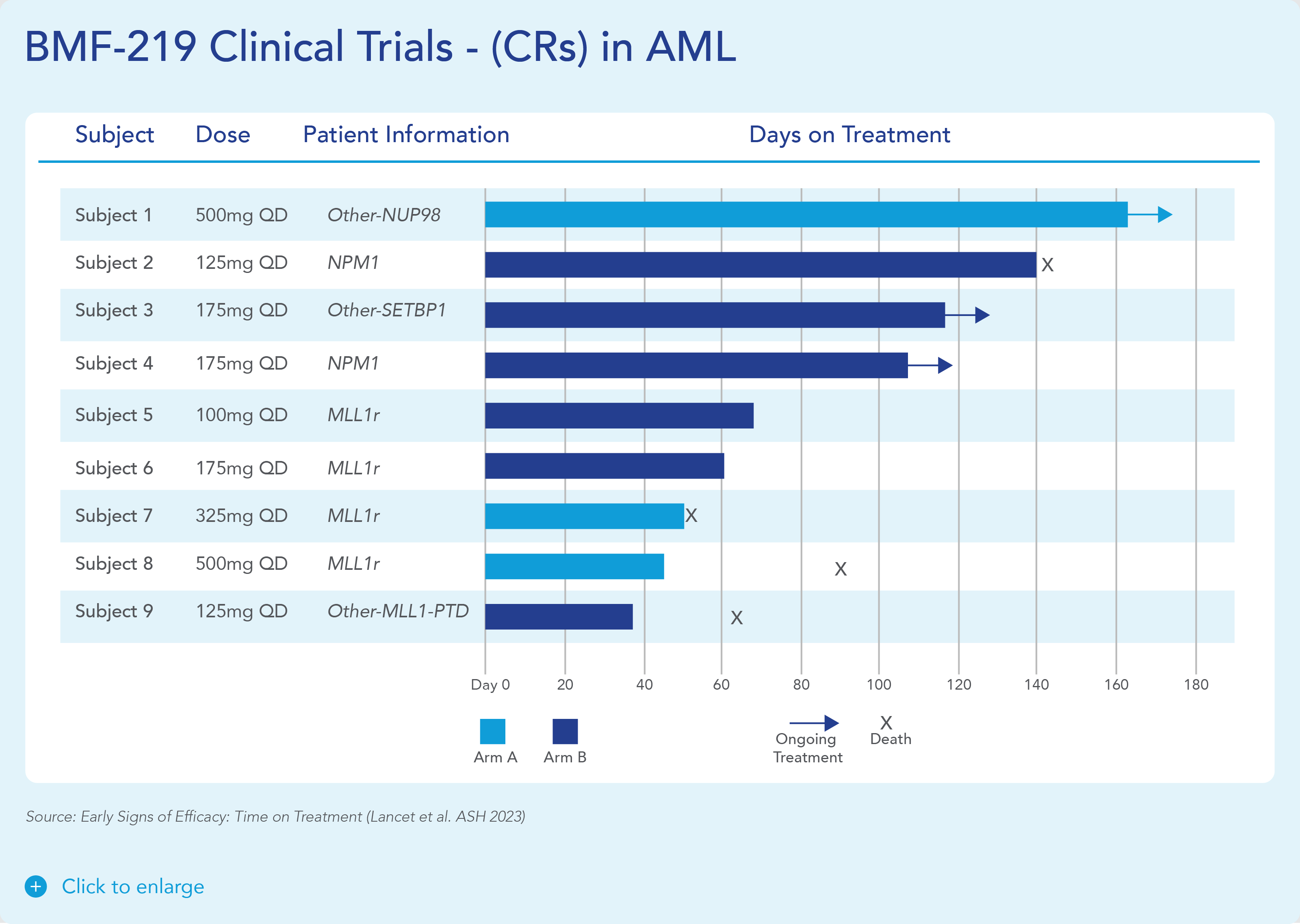
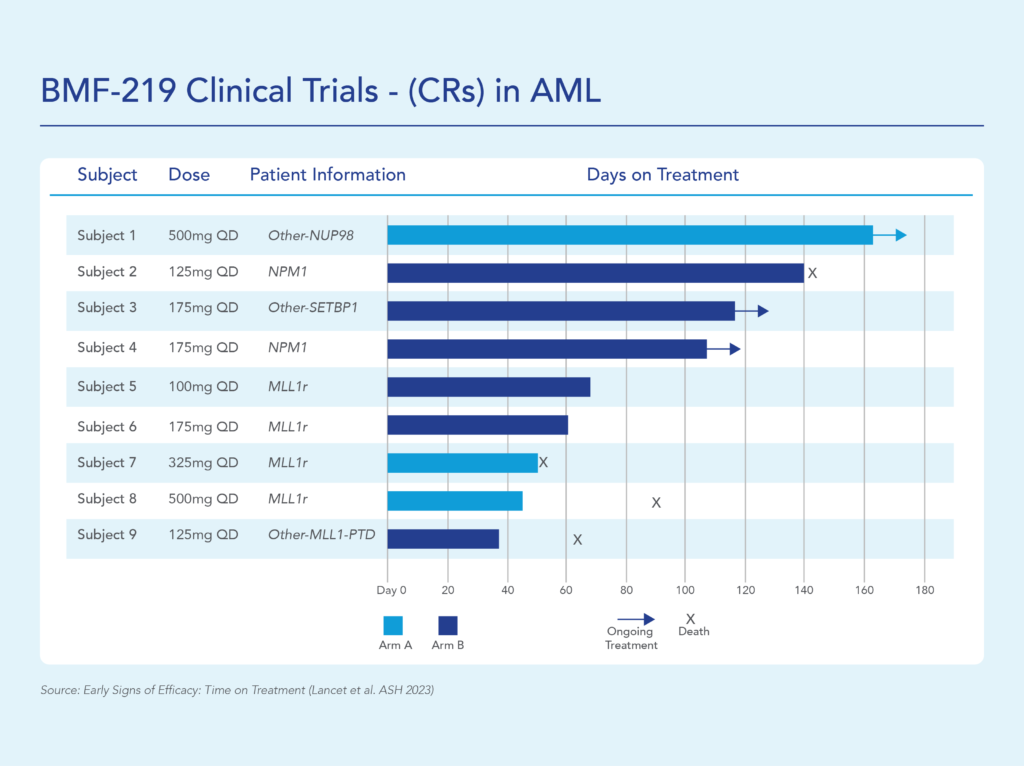
In December 2023, Biomea reported the achievement of minimal residual disease negativity (MRD-neg) in one of the first complete responses (CRs) in AML. Within the total of 7 AML patients who received at least 2 cycles of therapy by the cutoff date, 2 CRs were observed with a mean time to response of 1.8 months. Pharmacodynamic data from a case study of an AML patient containing NUP98-NSD1 mutation further supports the proposed mechanism of action of BMF-219 as a menin inhibitor; in-line with preclinical models, BMF-219 downregulated key leukemogenic genes (e.g., HOXA9, MEIS1) as well as MEN1.
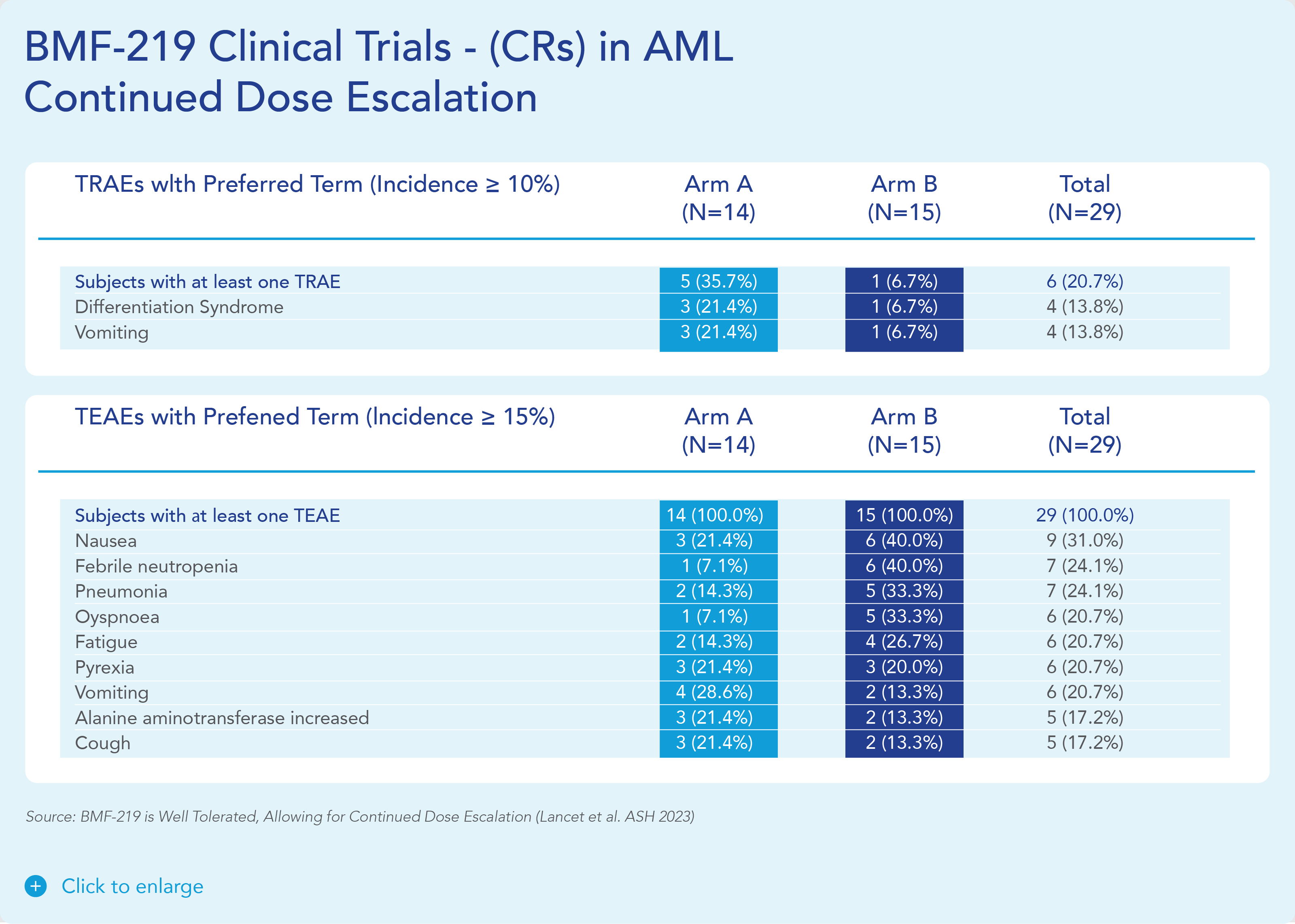
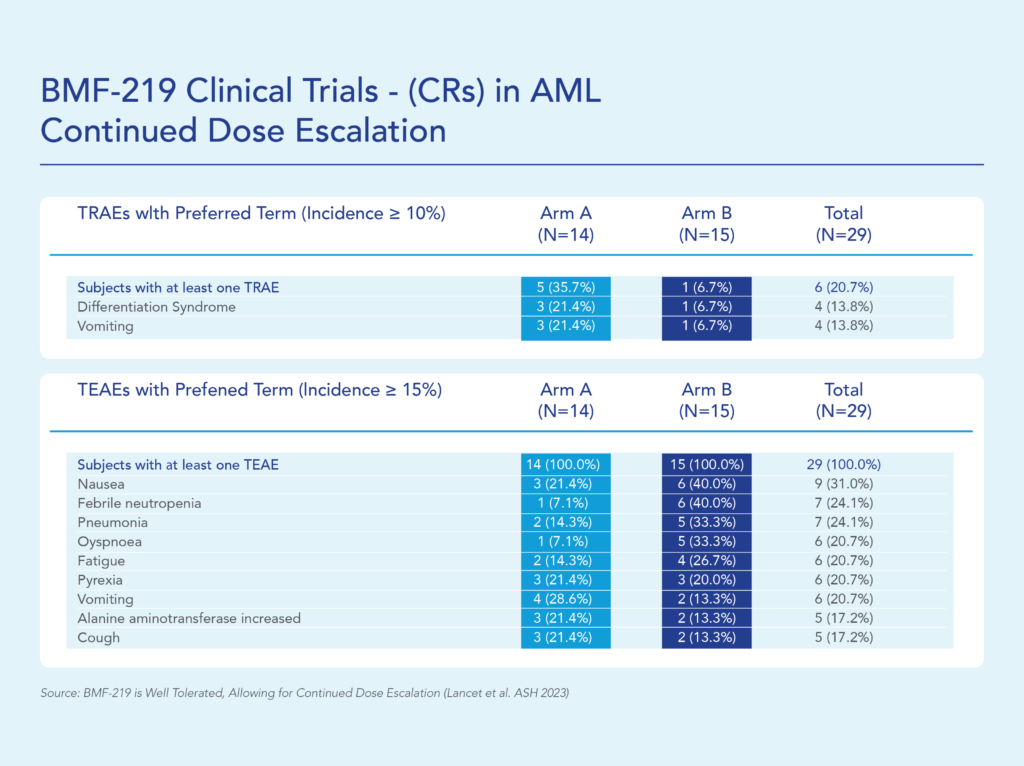
BMF-219 was generally well tolerated with no dose-limiting toxicities observed and without adverse event (AE)–related treatment discontinuations.
MLLr/mNPM1 Dependent AML/ALL
MLL1-r leukemias are characterized by MLL1 gene (encoded by the gene KM2TA) translocation abnormalities. These abnormalities result in the formation of fusion genes encoding fusion proteins comprised of MLL1 and a corresponding fusion partner domain. The interaction of these fusion proteins with menin drives the expression of downstream target genes such as HOXA9 and MEIS1, triggering leukemic cell proliferation.
Menin binds directly to the conserved N-terminus of MLL proteins, making it a promising target that could potentially be exploited consistently by a menin inhibitor therapeutic. Preventing the MLL proteins from binding to menin has been shown to abolish the oncogenic effects in vitro and in vivo.
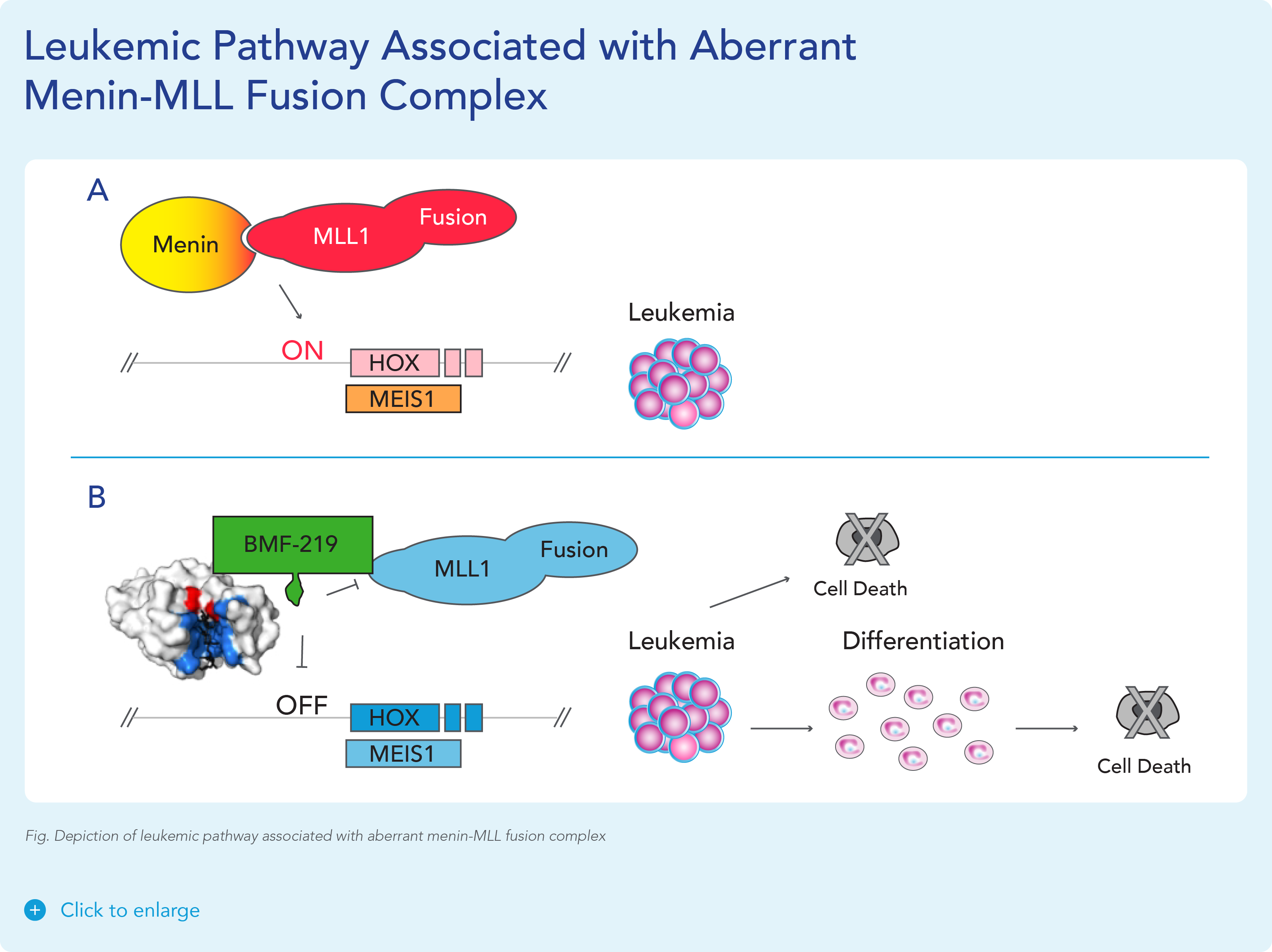
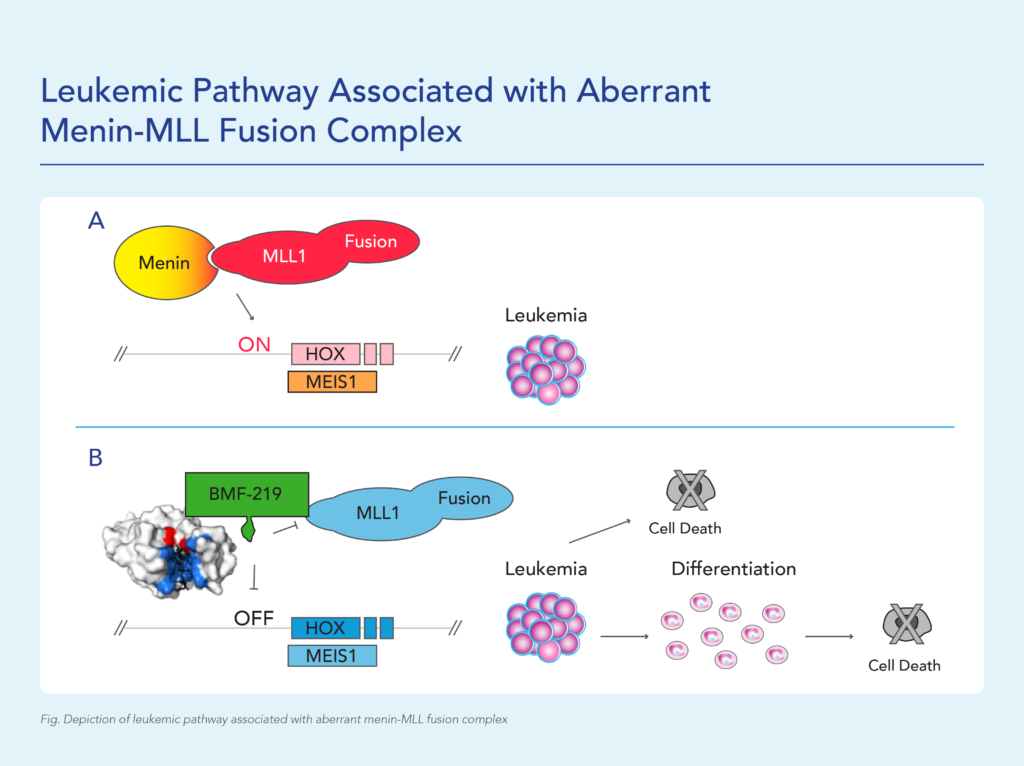
Approximately 20,000 and 6,000 patients in the United States are diagnosed annually with AML and ALL, respectively. KMT2Ar leukemia has limited therapeutic options and affects approximately 5-10% of acute leukemias in adults and approximately 70% of acute leukemias in infants. In addition to KMT2A, MLL signaling in some forms of MLL wild-type (MLL-wt) AML have also been implicated, including those bearing independent oncogenic mutations in nucleophosmin (NPM1), a molecular chaperone, and DNA-methyltransferase 3A (DNMT3A), a methyl transferase. These subpopulations together represent approximately 35-40% of AML cases.
A perhaps more dire area of unmet need is relapsed/refractory AML. Despite evolving insights into the pathogenesis of AML, over 11,000 patients with AML die each year from the disease in the United States. Relapse is the most common cause of treatment failure. The 5-year overall survival (OS) for adult patients with AML after disease relapse is only approximately 10%. Given the involvement of KMT2A and NPM1 in a high percentage of acute leukemias, and the poor clinical outcomes provided by available treatments, we believe a new treatment that can inhibit the function of both targets by disrupting or preventing interactions with menin could address this unmet need.
MYC Driven Liquid Tumors (Multiple Myeloma)
MYC is a transcription factor that is implicated in oncogenesis and typically regulates genes associated with cellular proliferation, differentiation, and apoptosis. In fact, MYC is constitutively and aberrantly expressed in over 70% of human cancers. MYC dysregulation can occur due to a number of different factors, including RAS activation. MYC appears to play a key role in the functioning of many cancer cells, including multiple myeloma (MM) and KRAS solid tumors (Colorectal, Pancreatic, and Lung). MYC is aberrantly expressed in Multiple Myeloma and is a downstream effector of KRAS mutant tumors.
Menin has been shown to play an essential role in the MYC transcriptional complex, which leads to menin-mediated enhancement of MYC target gene expression in cancer cells.
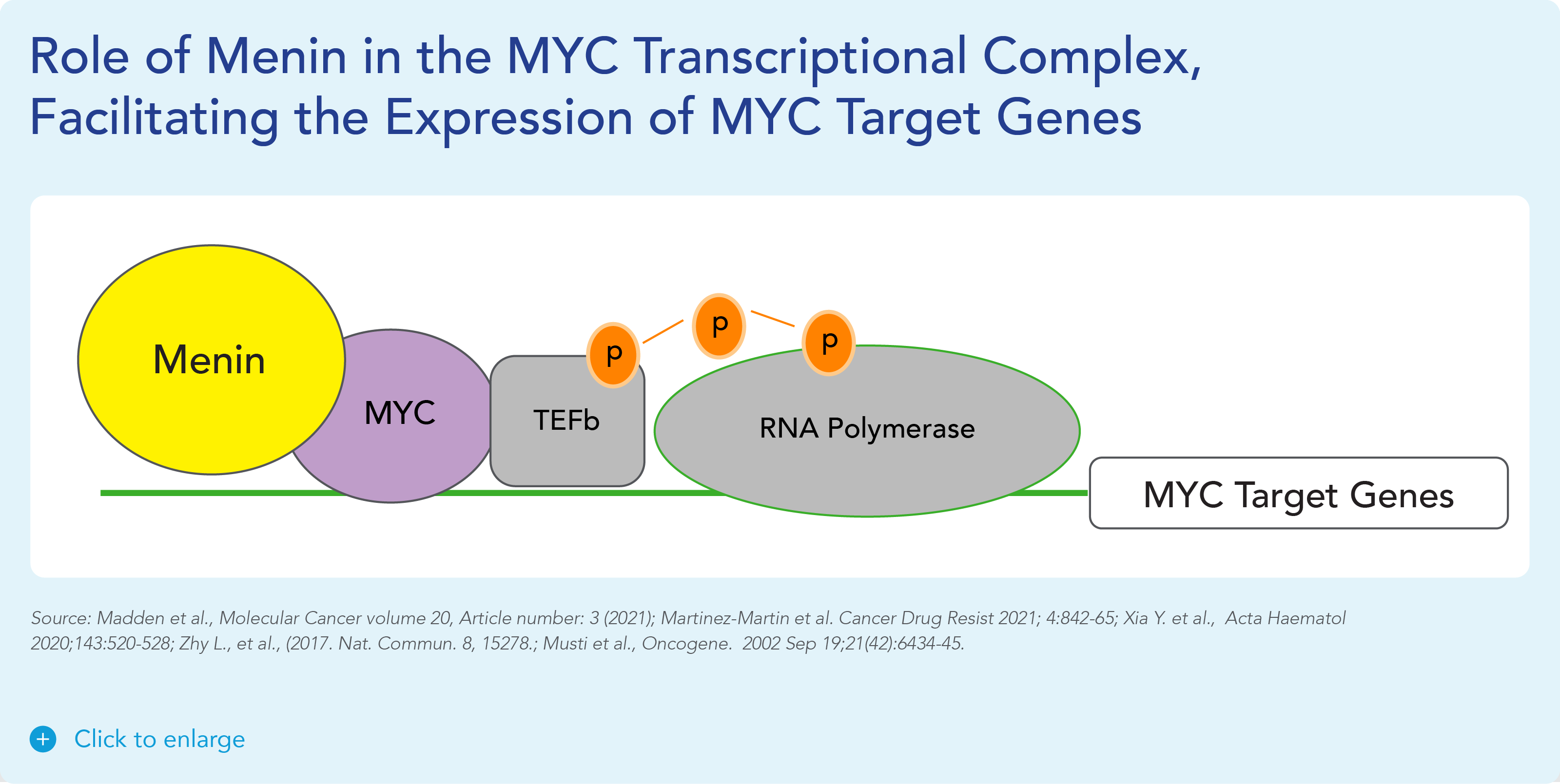
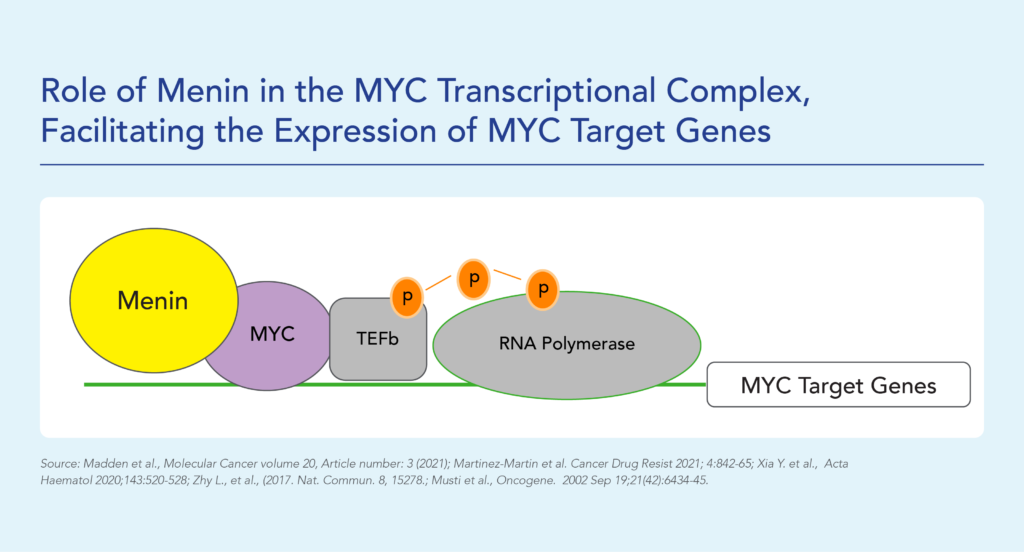
Fig. The role of menin in the MYC transcriptional complex, facilitating the expression of MYC target genes
BMF-219 demonstrated robust anti-tumor activity and mechanistic evidence for novel inhibition of the menin protein in preclinical models of MM.
MM is a cancer of plasma cells, which make antibodies and are mainly located in the bone marrow. Approximately 35,000 people in the U.S. are diagnosed with MM each year and the 5-year relative survival rate is ~56% (Source: NCI SEER Data). While many therapeutic options are available to patients, a subset of highly treatment-refractory patients exists. In these patients, overall survival is as low as 6 months. Additionally, it is estimated that more than 60% of MM patients have menin-dependent genetic drivers (MYC addicted or driven) and that these drivers are more common in the relapsed or refractory setting.
MYC Driven KRAS Solid Tumors (Lung, Colon, Pancreatic Cancer)
BMF-219 is the first menin inhibitor to enter clinical trials for the treatment of solid tumors. A targeted pan-KRAS inhibitor has the potential to treat 25-35% of NSCLC, 35-45% of CRC, and approximately 90% of PDAC patients. In a series of preclinical studies, BMF-219 showed strong and highly specific pan-KRAS anti-cancer activity as a single agent across KRAS G12C, G12D, G12V and G13D mutations, including in NSCLC, CRC, and the most prevalent type of pancreatic cancer, PDAC.
Non-Small Cell Lung Cancer (NSCLC) is the most common form of lung cancer, representing ~84% of all lung cancer cases or approximately 200,000 cases in the U.S. each year (Source: NCI SEER Data). Additionally, the five-year survival rate of NSCLC is ~25%. KRAS is a key node in the RAS signaling pathway and the most frequent oncogene in NSCLC, occurring in ~30% of patients with NSCLC.
Pancreatic ductal adenocarcinoma (PDAC) is a relatively rare form of cancer in the U.S., representing approximately 60,000 cases in the U.S. each year (Source: NCI SEER Data). It is an aggressive cancer with a very low five-year survival rate of ~11%, indicating that there is a large unmet need. It is rarely diagnosed early, contributing to the low survival rate. Among patients with pancreatic cancer, RAS mutations (including KRAS) occur in up to approximately 98% of patients.
Colorectal cancer (CRC) is the fourth most common form of cancer in the U.S., representing approximately 150,000 cases in the U.S. each year (Source: NCI SEER Data). These cancers start in the rectum or the colon and can be diagnosed/identified early, even potentially as noncancerous polyps. The five-year survival rate of CRC is approximately 65%. Among other mutations, KRAS mutations occur in approximately 50% of patients with CRC.
BMF-500 - FLT3 Inhibitor in AML
BMF-500 is an investigational, novel, orally bioavailable, highly potent and selective covalent small molecule inhibitor of fms-like tyrosine kinase 3 (FLT3). Biomea has continued advancing BMF-500 toward the clinic, with an investigational new drug (IND) application cleared in 1H2023 and a Phase 1 study of BMF-500 (COVALENT-103; NCT05918692) initiated and first patient dosed in 2H2023.
BMF-500 was discovered and developed in-house at Biomea using the company’s proprietary FUSION™ System and has demonstrated best-of-class potential based on extensive preclinical studies. The kinase profile of BMF-500 showed high target selectivity, suggesting the potential for minimal off-target liabilities. BMF-500 was designed to have a therapeutic profile to allow for combinations with standard of care and/or novel targeted agents like BMF-219.
FLT3 is a receptor tyrosine kinase (RTK) that plays a central role in the survival, proliferation, and differentiation of immature blood cells. Notably, FLT3 gene mutations are common in patients with AML and are associated with a poor prognosis. Nearly 30% of AML patients have a FLT3 mutation, representing more than 6,000 incident patients in the United States each year. While FLT3-specific and pan-tyrosine kinase inhibitors are approved by the FDA across various lines of therapy in AML, these agents have produced relatively low rates of durable responses, and overall survival remains an unmet need.
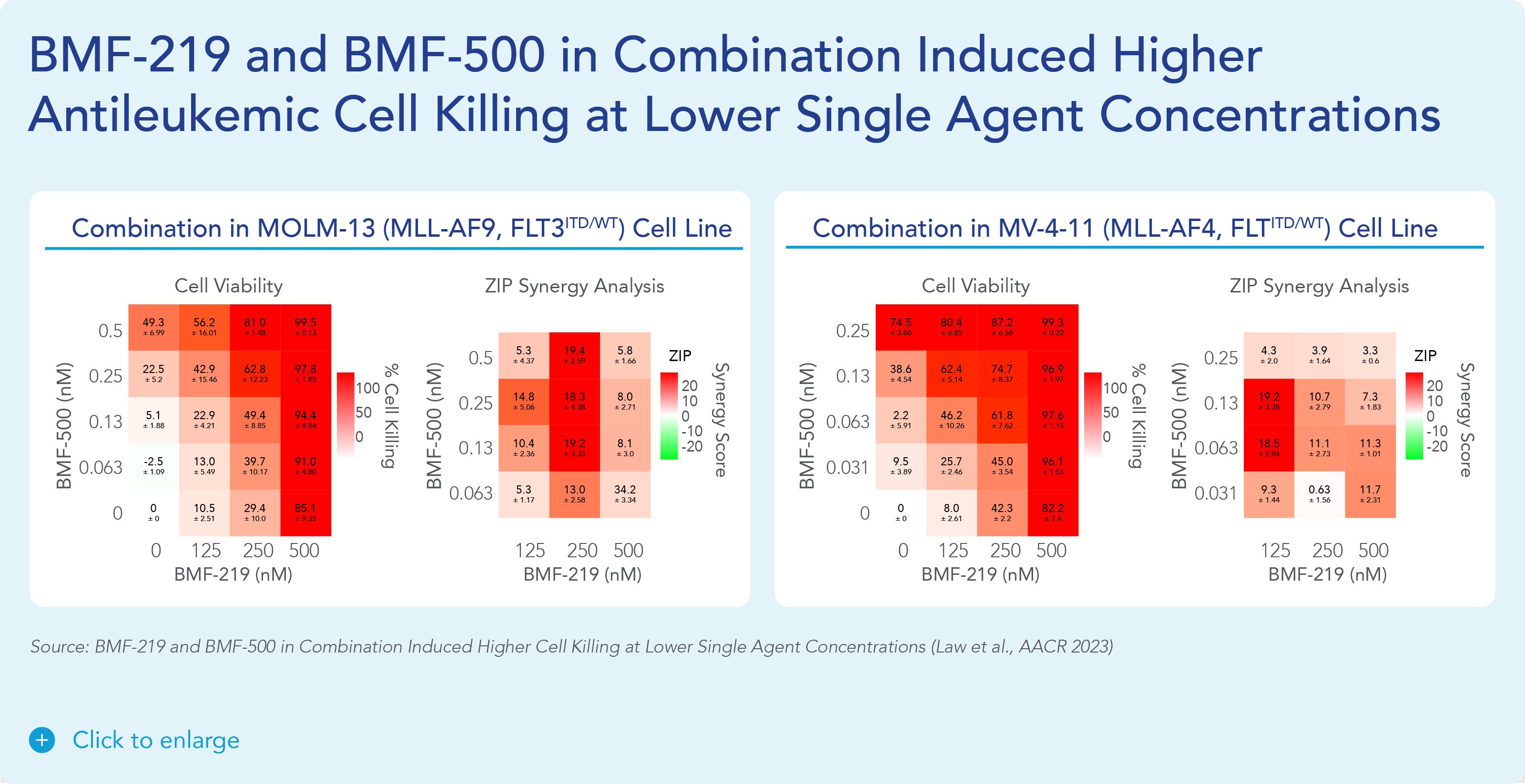
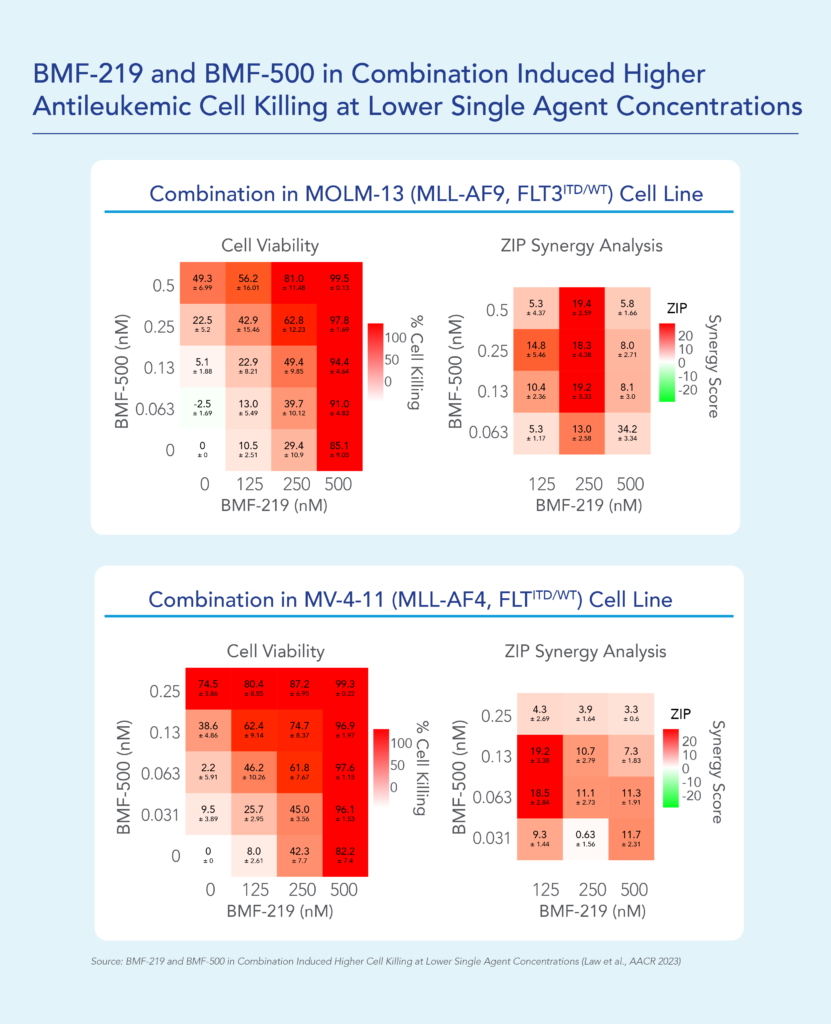
Previous data presented at medical conferences showed BMF-500’s picomolar affinity to activating FLT3 mutations, including FLT3-ITD and various tyrosine kinase domain (TKD) mutations (Law et al. ASH 2022). BMF-500 demonstrated multi-fold higher potency and increased cytotoxicity than commercially available non-covalent FLT3 inhibitor gilteritinib. Further data also exhibited the potential utility of combination strategies to achieve higher antileukemic cell killing with reduced concentrations of BMF-500 and BMF-219 (Law et al. AACR 2023). These data provide preclinical evidence for combining pathway-specific inhibitors as a promising therapeutic strategy for further investigation in acute leukemia.
Poster Presentations
BMF-219 was discovered and developed in-house at Biomea using the company’s proprietary FUSION™ System. BMF-219 is a covalently binding inhibitor of menin, a protein known to play an essential role in oncogenic signaling in genetically defined leukemias as well as in diabetes. Preclinically, BMF-219 has demonstrated in well-established acute leukemia cell lines robust downregulation of key leukemogenic genes in addition to menin itself. Additionally, BMF-219 has shown anticancer activity in multiple in vitro, in vivo, and ex vivo models of acute leukemia, multiple myeloma, diffuse large B-cell lymphoma, and chronic lymphocytic leukemia. BMF-219 is currently being evaluated in clinical trials enrolling patients with specific menin-dependent mutations in liquid and solid tumors.
Literature References
The literature references section of Biomea Fusion serves as a comprehensive repository of scientific and clinical research that underpins the company’s innovative work in type 1 and type 2 diabetes, precision oncology and beyond. This collection highlights key studies and publications that inform and validate Biomea’s approach to targeting diseases through covalent small molecules. By curating these references, Biomea Fusion emphasizes its commitment to evidence-based research and collaboration with the scientific community.
Covalent Inhibition
The article explores the emergence and increasing importance of targeted covalent inhibitors in drug discovery. It discusses how these inhibitors are designed to form durable bonds with specific target proteins, leading to enhanced potency and selectivity compared to traditional non-covalent inhibitors. The review covers key principles in the design and optimization of targeted covalent inhibitors, as well as their applications across various disease areas.
The article addresses the challenges associated with targeting PPIs, which are crucial for cellular signaling and often implicated in cancer progression. It discusses various approaches and techniques employed in the design of covalent PPI inhibitors, emphasizing the need for specificity and potency. Case studies and examples are provided in preclinical and clinical settings, highlighting their potential in overcoming resistance mechanisms and enhancing treatment efficacy in cancer.
This perspective article discusses fundamental concepts such as the mechanisms of covalent binding between drugs and their targets, as well as factors influencing the kinetics of these interactions. The author explores how understanding the kinetic properties of covalent and irreversible inhibitors can inform drug design and optimization processes. The article also addresses the implications of kinetic parameters on efficacy, selectivity, and safety profiles of covalent drugs.
The article discusses the advantages of covalent drugs regarding enhancing potency and selectivity. It highlights the challenges in designing covalent drugs, such as ensuring specificity and minimizing off-target effects to minimize potential toxicity. The review also covers various strategies for optimizing covalent drug candidates, including medicinal chemistry approaches and the use of advanced screening techniques.
Menin Science
The article discusses the dynamics and functional insights of menin, a protein implicated in various cellular processes. It delves into the molecular interactions and regulatory roles of menin, particularly its partnerships with other proteins and its impact on cellular functions. The study highlights how menin’s interactions influence gene expression and signaling pathways, emphasizing its role as a crucial regulator in cell growth and differentiation.
The study delves into how menin, a protein encoded by the MEN1 gene, regulates various aspects of hematopoietic stem cell differentiation and proliferation. The authors discuss studies that highlight menin’s involvement in maintaining hematopoietic stem cell homeostasis and its impact on lineage commitment towards specific blood cell types. They also examine the consequences of MEN1 gene mutations on hematopoietic function, linking these alterations to diseases like multiple endocrine neoplasia type 1 (MEN1) syndrome.
The study delves into how menin, a protein encoded by the MEN1 gene, regulates various aspects of hematopoietic stem cell differentiation and proliferation. The authors discuss studies that highlight menin’s involvement in maintaining hematopoietic stem cell homeostasis and its impact on lineage commitment towards specific blood cell types. They also examine the consequences of MEN1 gene mutations on hematopoietic function, linking these alterations to diseases like multiple endocrine neoplasia type 1 (MEN1) syndrome.
The study demonstrates that insulin plays a significant role in regulating menin expression, cytoplasmic localization, and its interaction with FOXO1 (Forkhead Box O1), a transcription factor involved in insulin signaling. The study reveals that insulin stimulation leads to increased menin expression and promotes its translocation from the nucleus to the cytoplasm, where it interacts with FOXO1. This interaction is critical in mediating insulin’s effects on gene expression and cellular responses related to glucose homeostasis and insulin sensitivity.
In the review, Menin is highlighted as a key regulator influencing various cellular processes essential for liver and bile duct function, including transcriptional regulation, cell cycle control, and DNA repair mechanisms. The review emphasizes menin’s interaction with numerous transcription factors and chromatin-modifying enzymes, underscoring its ability to modulate gene expression critical for hepatobiliary homeostasis. The authors discuss the implications of MEN1 gene mutations, which are implicated in the pathogenesis of primary liver cancers and bile duct disorders. They explore menin’s involvement in signaling pathways crucial for liver development and its responses to pathological stimuli.
The review examines the role of the menin pathway in epigenetic regulation. Menin, encoded by the MEN1 gene, serves as a scaffold protein that interacts with histone modifiers to regulate chromatin structure and gene expression. Dysregulation of the menin pathway, often due to MEN1 mutations, disrupts these epigenetic mechanisms, contributing to tumorigenesis and cancer progression.
Menin in Hematological Malignancies
In acute leukemias, dysregulation of menin contributes to leukemogenesis through interactions with key oncogenic pathways such as MLL fusion proteins. Preclinical studies using menin inhibitors have shown promising results in disrupting leukemic cell growth and enhancing sensitivity to chemotherapy. Clinical trials investigating menin inhibitors as targeted therapies for acute leukemias, particularly in combination with standard treatments, are ongoing. The review highlights the potential of menin inhibition as a novel therapeutic strategy in the management of acute leukemias, providing insights into future directions for research and clinical applications.
The article discusses challenges including the development of small molecule inhibitors that selectively disrupt the menin–MLL interface without affecting normal cellular functions of menin. The review discusses various strategies and structural insights into designing effective menin inhibitors, highlighting the importance of understanding the molecular basis of the interaction. The article emphasizes ongoing research efforts and the future directions needed to optimize menin–MLL interaction inhibitors for clinical use.
The article examines MYC alterations in DLBCL, encompassing rearrangements, amplifications, and mutations that drive dysregulated MYC expression, fostering tumor growth and resistance. It details the prevalence and prognostic impact of these alterations in DLBCL, correlating them with aggressive disease behavior and poorer patient outcomes. The review highlights challenges in identifying and characterizing MYC alterations, advocating for standardized diagnostic methods and therapies targeting MYC pathways to improve DLBCL management.
The article explores the therapeutic targeting of MYC in MM. MYC dysregulation contributes to MM pathogenesis by promoting cell proliferation and survival. The review discusses various strategies to inhibit MYC, including direct targeting with small molecules and indirect approaches via upstream regulators. It highlights the challenges associated with targeting MYC due to its complex regulatory network and emphasizes the potential of combination therapies to overcome resistance mechanisms.
The study investigates targeting chromatin regulators to inhibit leukemogenic gene expression in NPM1 mutant leukemia. They demonstrate that NPM1 mutations lead to altered chromatin structure and enhanced transcriptional activation of oncogenic genes. By using small molecule inhibitors targeting chromatin regulators such as BRD4 and DOT1L, they effectively suppress the expression of these leukemogenic genes. The research highlights the potential therapeutic strategy of epigenetic modulation to specifically target and disrupt the oncogenic pathways in NPM1 mutant leukemia.
Menin in Solid Tumors
The article explores how the same binding pocket in menin interacts with both MLL and JUND proteins, but exerts opposite effects on transcriptional regulation. Menin forms a complex with MLL, promoting histone H3 lysine 4 (H3K4) methylation and activation of target genes involved in leukemogenesis. In contrast, its interaction with JUND inhibits transcription by disrupting the assembly of RNA polymerase II preinitiation complex. This dual role of menin highlights its context-dependent function in gene expression regulation, influencing both activation and repression of target genes critical in cellular processes and disease pathogenesis.
The study identifies JunD, rather than c-Jun, as the critical AP-1 transcription factor necessary for Ras-induced lung cancer. Through genetic mouse models and human lung cancer cell lines, the research demonstrates that JunD depletion significantly impairs Ras-driven tumorigenesis in the lung. JunD deficiency attenuates tumor growth and reduces malignant characteristics such as proliferation and invasive potential. These findings underscore JunD as a potential therapeutic target in Ras-driven lung cancer, highlighting its pivotal role in promoting oncogenic signaling pathways and tumor progression in this context.
The study investigates the role of MLL in regulating Rasgrf1 and its impact on Kras-driven lung tumorigenesis. They demonstrate that loss of MLL leads to epigenetic dysregulation of Rasgrf1, a gene implicated in Ras signaling pathways. In mouse models and human lung cancer cells, MLL deficiency attenuates Kras-induced lung tumor formation by decreasing Rasgrf1 expression, thereby inhibiting downstream signaling cascades crucial for tumor growth. The findings suggest that MLL plays a critical role in maintaining Rasgrf1 expression levels through epigenetic mechanisms, highlighting its potential as a therapeutic target for Ras-driven lung cancers.
The study demonstrates that menin interacts with c-Myc to facilitate its transcriptional activity, thereby promoting cell proliferation and survival in cancer cells. Through a series of molecular experiments and genomic analyses, the authors reveal that menin depletion attenuates c-Myc-driven oncogenic pathways, leading to reduced tumor growth and aggressiveness in various cancer models. The study underscores menin as a critical regulator of c-Myc function in cancer cells, highlighting its potential as a therapeutic target for cancer treatment strategies aimed at disrupting oncogenic signaling pathways mediated by c-Myc.
The study demonstrates that menin is crucial for activating the MYC locus and promoting MYC-mediated transcriptional activity in this context. Through experimental approaches, including genetic and biochemical analyses, the study reveals that menin depletion attenuates MYC-driven signaling pathways and reduces androgen receptor transcriptional output, leading to impaired proliferation and survival of prostate cancer cells.