About
About
Biomea Fusion
We are a clinical-stage diabetes and obesity medicines company focused on the development of our oral small molecules, icovamenib and BMF-650, both designed to significantly improve the lives of patients with metabolic disease. We believe that we have found a method to reverse the root cause of diabetes leading to reduced dependency on life-long chronic therapy.
Our lead development candidate, icovamenib, is positioned to be the first disease modifying treatment for diabetes. Icovamenib inhibits menin, which thereby promotes restoration of insulin-producing beta cells, which are depleted by approximately 50% at the time of diagnosis of Type 2 Diabetes.1 The functional restoration of this replenished pool of beta cells can potentially provide significant glycemic benefits for diabetes patients.
Icovamenib’s mechanism has also shown synergistic effects when combined with a GLP-1-based therapy, allowing for enhanced efficacy in glycemic management and weight reduction, additionally paired with preservation of lean muscle mass.
Our second development candidate, BMF-650, is a next-generation oral GLP-1 receptor agonist similar to the Chugai/orforglipron chemotype class, optimized for improved properties and projected to enter clinical studies in the second half of 2025.
With our programs, we are focused on addressing metabolic disorders, a public health concern impacting approximately 50% of Americans and 20% of the world’s population. We aim to cure.
Reference: 1 Diabet Med 1988; 5 :36 –41

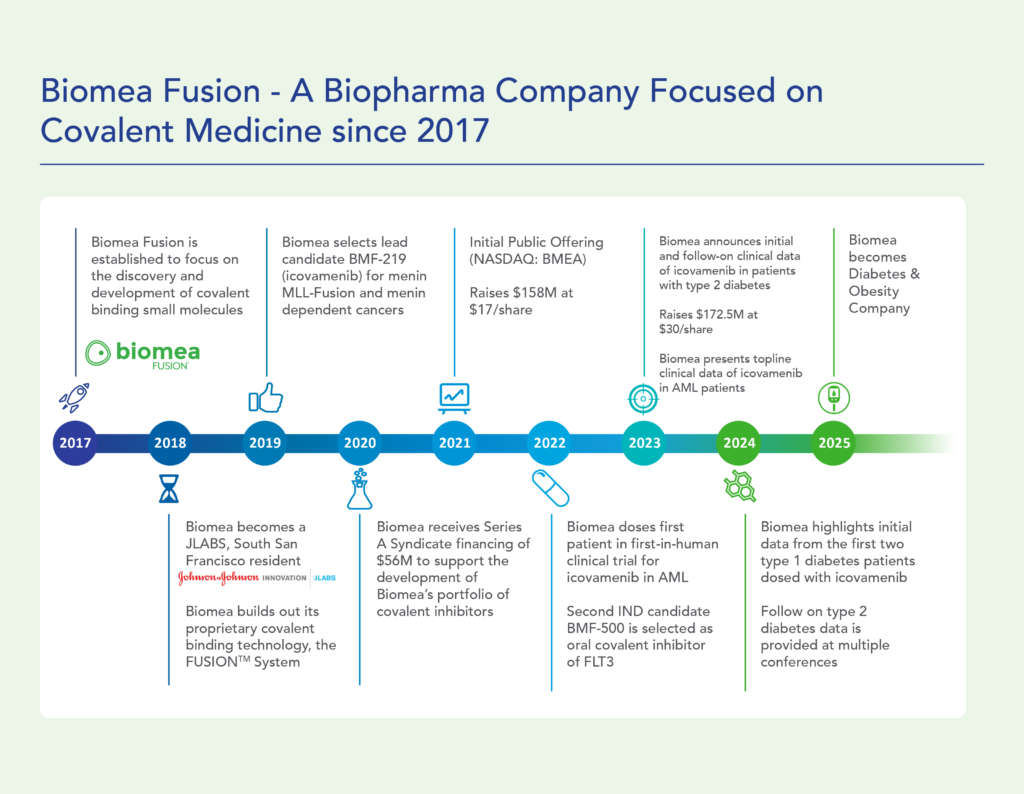
Biomea Fusion, Inc. was formed in 2017 by a small group of biotech executives. We came together after a career in drug design and development with the common goal of achieving better therapies for patients.